![Basic concepts: Acid-Base chemistry & pH 1.Recognizing acid/base and conjugate base/acid 2.Calculation of pH, pOH, [H 3 O + ], [OH - ] 3.Calculating pH. - ppt download Basic concepts: Acid-Base chemistry & pH 1.Recognizing acid/base and conjugate base/acid 2.Calculation of pH, pOH, [H 3 O + ], [OH - ] 3.Calculating pH. - ppt download](https://images.slideplayer.com/15/4572591/slides/slide_45.jpg)
Basic concepts: Acid-Base chemistry & pH 1.Recognizing acid/base and conjugate base/acid 2.Calculation of pH, pOH, [H 3 O + ], [OH - ] 3.Calculating pH. - ppt download

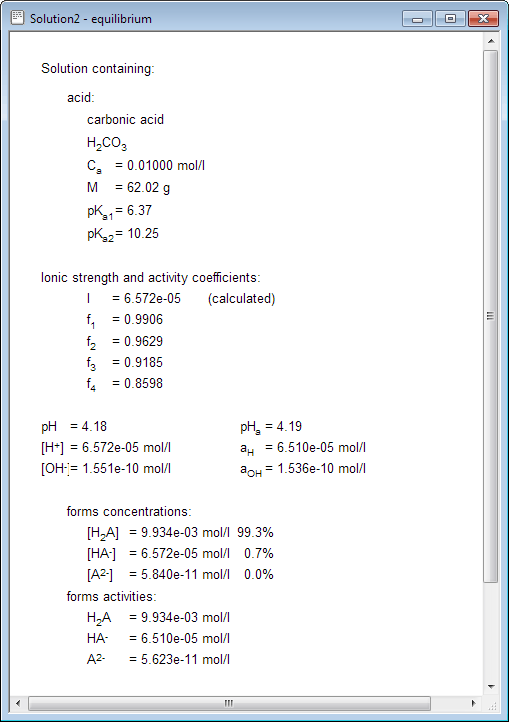
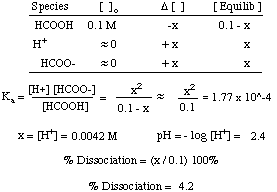
SOLVED: The pH of a solution depends upon the concentration of the acid (or base) as well as the extent of the reaction (2) as given by the equilibrium constant K: The