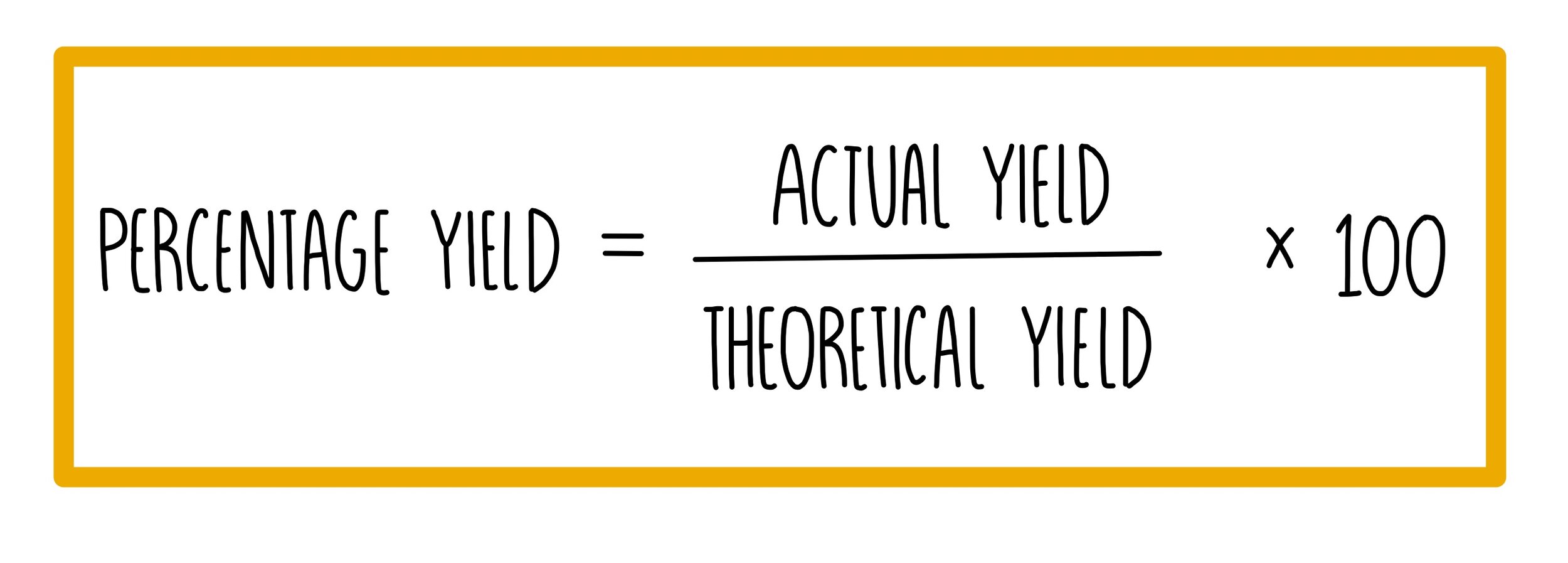
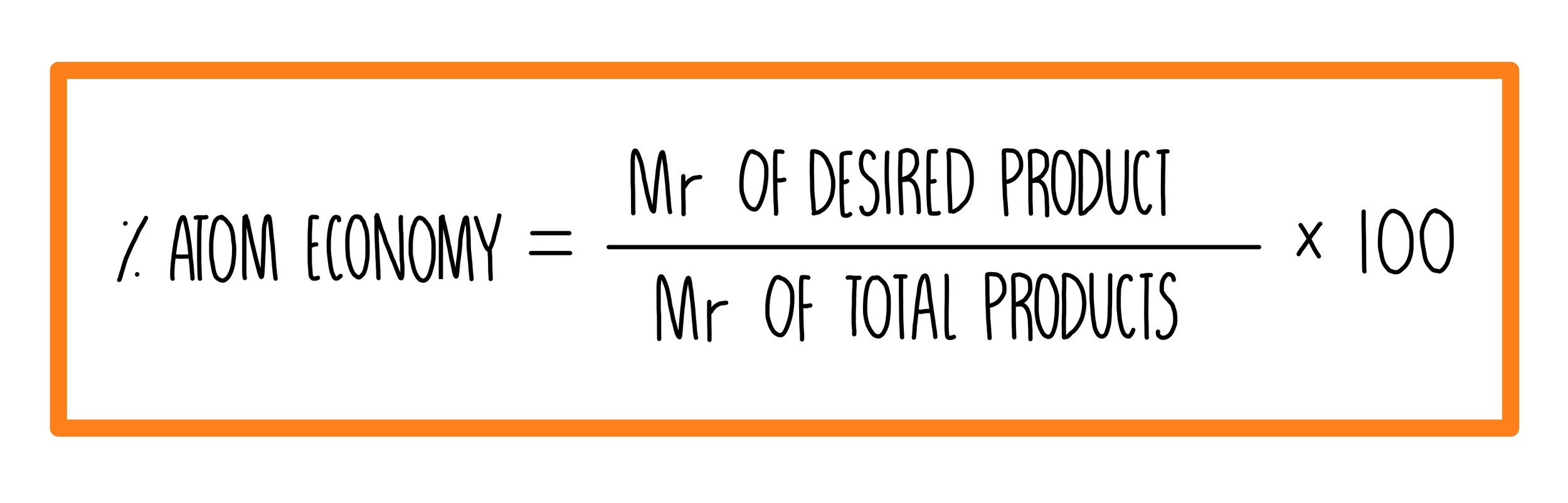
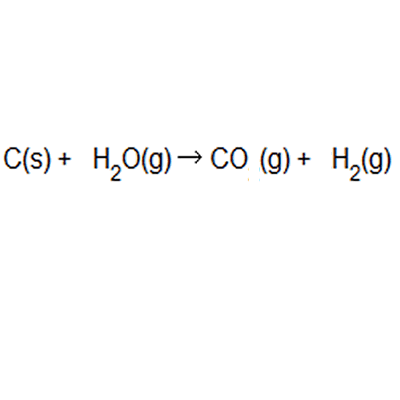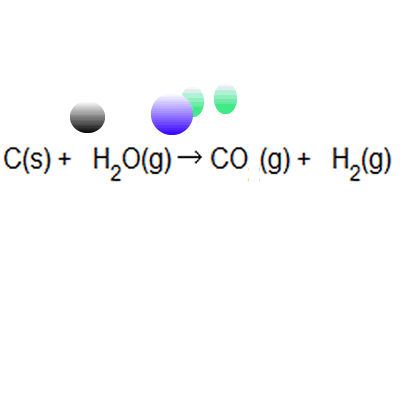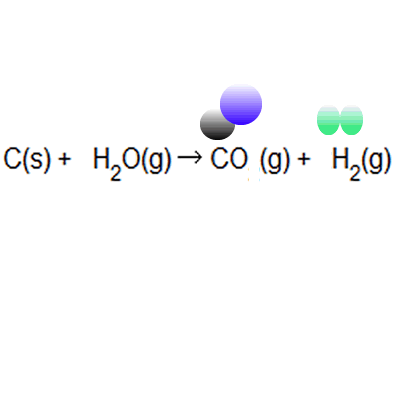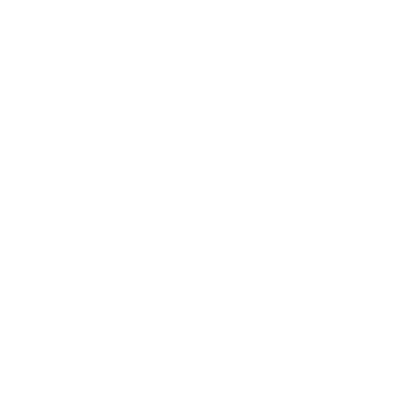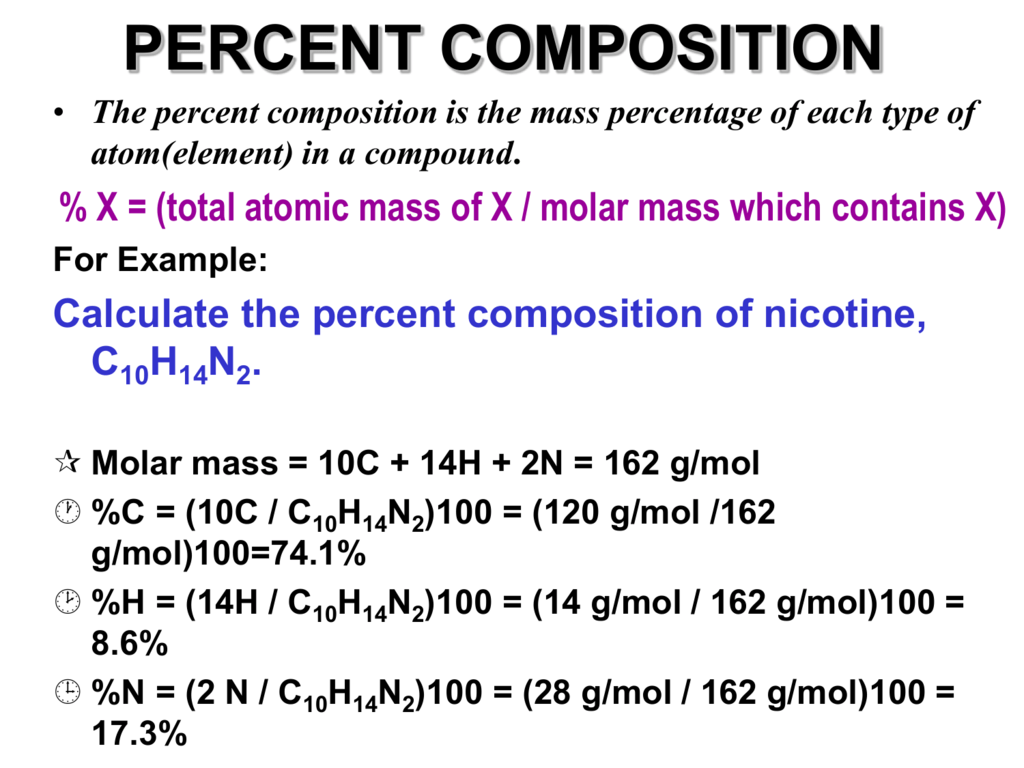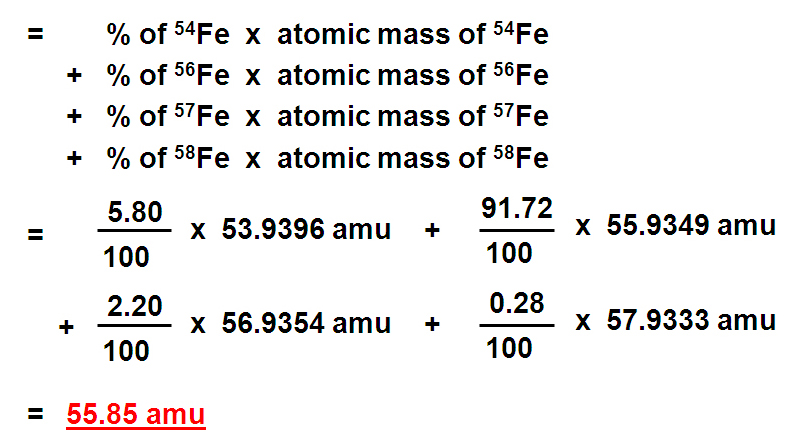Naturally occurring boron consists of two isotopes whose atomic weight are 10.01 and 11.01 . The atomic weight of the natural boron is 10.81 . Calculate the percentage of each isotopes in natural boron.
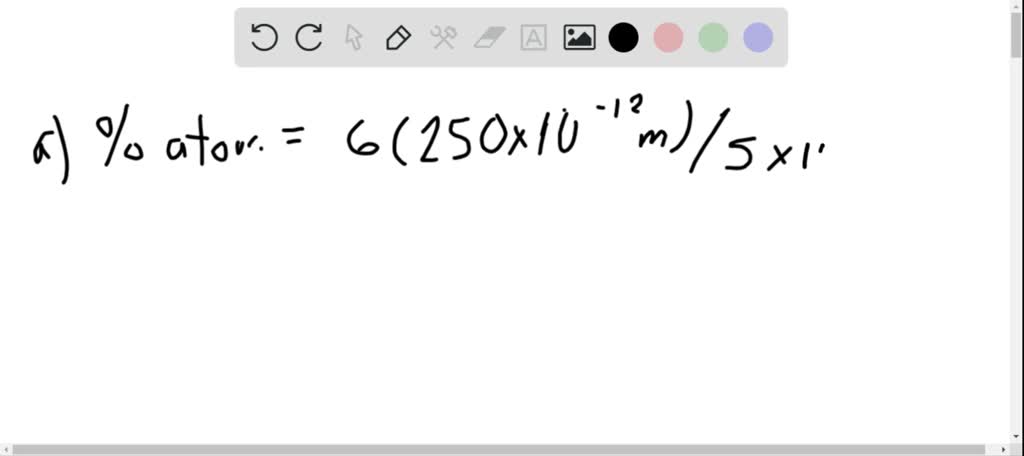
SOLVED:Calculate the percentage of the atoms that are on the surface of a cubic nanoparticle if the diameter of the atoms is 250 pm and the edge length of the particle is (